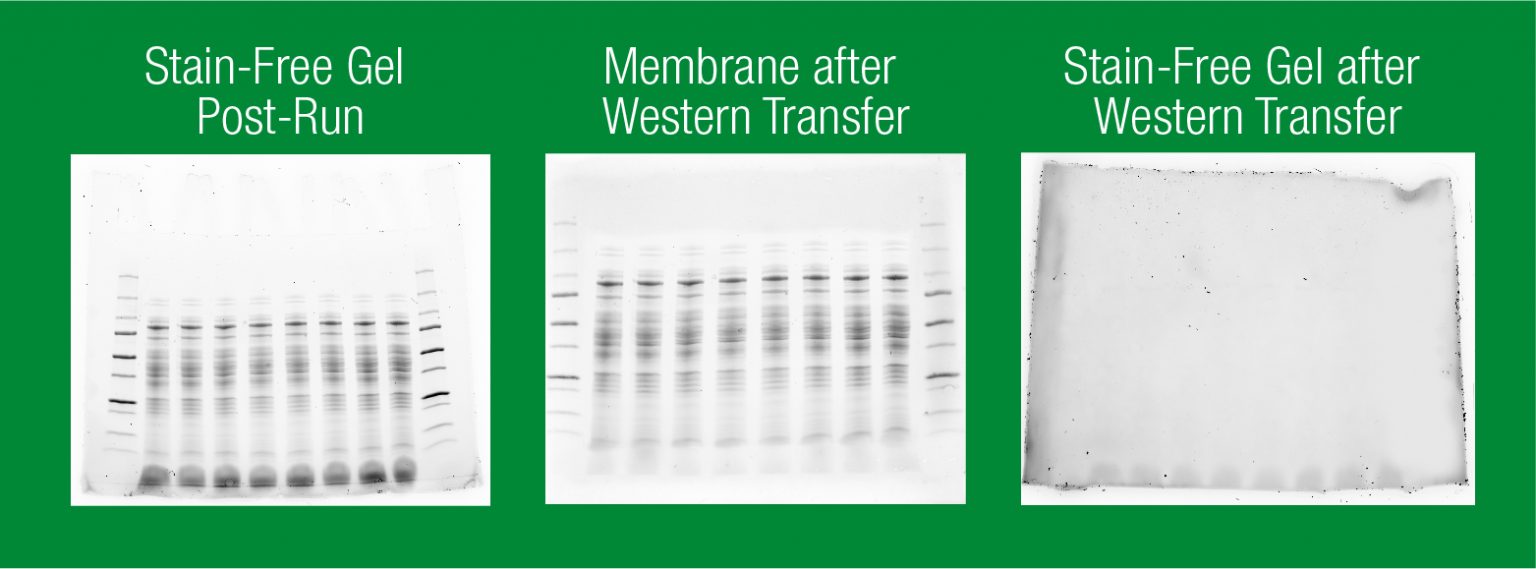
Check the output capacity of the power supply.Make sure the voltage and current output of the power supply match the needs of the blotting instrument.The power supply circuit is inoperative or an inappropriate power supply was used Reduce or eliminate the alcohol in the transfer buffer.SDS can increase transfer efficiency but it can also reduce binding efficiency to nitrocellulose and affect reactivity of some proteins with antibodies A protein near its isoelectric point (pI) will transfer poorly (buffer pH should be 2 pH units higher or lower than the pI of the protein of interest for optimal transfer efficiency) Use a more basic or acidic transfer buffer to increase protein mobility.The charge-to-mass ratio is incorrect (native transfers) Check the polarity of the connections to the power supply.Check the assembly of the transfer cell.Check the gel/membrane sandwich assembly.Proteins moved in the wrong direction (the gel/membrane sandwich may have been assembled in the wrong order, the cassette inserted in the tank in the wrong orientation, or polarity of the connections may be incorrect) Prepare fresh transfer buffer (never reuse transfer buffer).Transfer buffer was incorrect or prepared incorrectly Power conditions were too high or transfer time too long (proteins may transfer through the membrane and into the filter paper) (Do not do this if using Trans-Blot® Turbo system) Equilibrate gel in transfer buffer for 10 minutes prior to transfer.If an incorrect power supply is used, it is possible to not reach the set voltage if the current of the power supply is at its maximum limit Use a power supply with a high current limit.Check the current at the beginning of the run it may be too low for a particular voltage setting, indicating incorrect buffer composition.Increase the transfer time (thicker gels require longer transfer times).Power conditions were inadequate or transfer time too short Alternatively, one could use stain-free technology and LF PVDF membranes For example, stain the gel with Bio-Safe™ Coomassie or SYPRO Ruby stain, and stain the blot with Ponceau S stain). Poor electrophoretic transfer bands appear weak on blot (ensure proteins have been transferred by staining both the gel and blot with a total stain. Isoelectric Focusing Gels, Native Gels, Basic Proteins, and Acid-Urea Gels (0.7% acetic acid) Guide to power settings for different gel types. Cooling is generally required for all high-intensity transfers (except when using the Trans-Blot ® SD cell) and is recommended for long, unsupervised runs. The values presented in the table are guidelines - transfer conditions should be optimized for every transfer application. Transfer times are increased for gradient gels and decreased for low molecular weight proteins. The table below provides general guidelines for the voltage and current settings recommended for selected gel and buffer systems. Different transfer apparatuses, when used with different gel and buffer systems, require different power settings. antibody, antigen, technique, or buffer related where applicable. Therefore, the potential causes and solutions have been organized in this manner, and also with regard to potential problem sources, i.e. In most cases, the rest of the troubleshooting issues can be grouped into three major types: no bands, faint bands, and signal on Western blots that interferes with bands. Since one issue that commonly arises during Western blotting is the presence of unusual or unexpected bands on the blot, our troubleshooting section begins with a table describing some common reasons and potential solutions for addressing and evaluating this type of problem. Īdditionally, the use of negative and positive controls are of great help in assessing where things have gone wrong, and eliminating some possible sources of error under consideration. As mentioned at the end of Chapter 3, the proper antibody concentration can be determined with the use of dot blots and slot blots, significantly reducing the need for future troubleshooting. 
Many of these problems can be avoided with careful attention to experimental protocols, and with optimization at key stages throughout the procedure. In this chapter, suggestions are offered to assist in identifying and resolving some frequently encountered problems that arise during the course of Western blotting. When this occurs, it is useful to be able to quickly isolate the possible causes and to formulate an effective solution by troubleshooting the experiment. While Western blotting is a relatively simple and straightforward technique, it does not always yield results that meet with expectations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
